
82
Essential oil of Cannabis sativa L. strains
Vito Mediavilla and Simon Steinemann
Swiss Federal Research Station for
Agroecology and Agriculture, Reckenholzstrasse 191, 8046 Zurich, Switzerland
(E-mail: vito.mediavilla@fal.admin.ch
http://www.admin.ch/sar/fal/).
Mediavilla, Vito and Simon Steinemann 1997. Essential oil of Cannabis sativa L. strains. Journal of the International Hemp Association 4(2): 80 - 82. The aroma of hemp (Cannabis sativa L.) could be of considerable commercial value if evaluation of varieties and development of extraction methods led to a pleasing scent in the resulting essential oils. We compared the composition and smell of some fiber hemp and drug Cannabis essential oils isolated by steam distillation. The essential oil of some hemp strains contained particular monoterpenes and sesquiterpenes that imparted to the specimen a desireable scent. These preliminary one-year results do not take into account the influence that harvest time and the weather "just-before-harvest" could have on the quality of the essential oil. The Δ9-tetrahydrocannabinol (THC) concentration in the essential oils was very low and varied between 0.02% and 0.08%. The ratio of this compound to cannabidiol showed only small changes during steam distillation.
Introduction
The Cannabis smell is a peculiarity of this plant.
Its aroma does not originate from the terpenophenolic cannabinoids, but from the
more volatile monoterpenes and sesquiterpenes (Lehmann 1995). Hashish detection
dogs, for example, do not smell Δ9-tetrahydrocannabinol
(THC) but are able to smell the sesquiterpene caryophyllene oxide (Stahl and
Kunde 1973). According to Turner et al.
(1980) 58 monoterpenes and 38 sesquiterpenes have been identified in hemp. Using
steam distillation, it is possible to concentrate most of these components to an
essential oil.
Many
utilizations for hemp essential oil are known. They impart the typical Cannabis
aroma to such products as cosmetics, soaps, shampoos, creams, oils, perfumes
and also to foodstuffs. Additional possible uses are for aroma therapy and as a
means for plant protection. According to McPartland (1997), two essential hemp
oil components (limonene and alpha-pinene) have a repellent effect
against many insects. The bacteriostatic activity of hemp essential oil has been
reported by Fournier et al. (1978). Although first trials of hemp
essential oil used against potato late blight (Phytophthora
infestans) were not promising (Krebs 1996),
cannabinoid antifungal activity cannot be discounted.
The aim
of the work presented here was to assess the variability of hemp essential oil
from different Cannabis strains.
Materials and methods
Fiber and drug cultivars (Tab. 1) were grown in 1996 near
Zurich, Switzerland (approximately 47ş 25’ N, 8ş 30’ E, 400 m elevation).
The crop was harvested between the end of flowering and seed ripeness. Flowers
and the upper leaflets of female or hermaphrodite plants were cut by hand and
freshly distilled. Steam distillation in a copper still with 0.5 kg plant
material took 30 minutes. The essential oil was collected using a
lighter-than-water volatile oil apparatus consisting of a glass funnel.
Monoterpene and sesquiterpene analyses were carried out by GC/MS, and
cannabinoid analyses by GC alone.
Scent tests were performed with 15
volunteers who took part in smelling hemp essential oils diluted with jojoba oil
(1: 5).
Table 1. Hemp strains and harvest stage.

Results
The yield of hemp essential oil amounted to approximately
1.3 liter/ton fresh weight, which corresponds to about 10 liters per hectare. No
quantitative yield assessment was done.
We could
characterize 16 terpenoid compounds in the essential oil of different Cannabis
strains (Tab. 2). The concentration of monoterpenes was generally higher
than that of sesquiterpenes, varying from 47.9% to 92.1% of total terpene
content. Sesquiterpene concentrations varied from 5.2% to 48.6%. The most
abundant substance was myrcene, followed by trans-caryophyllene, alpha-pinene,
trans-ocimene
and alpha-terpinolene. The composition of the different essential oils
varied greatly. For example, the oil of strain B 3985 TE was rich in alpha-pinene,
beta-pinene and limonene concentration, ‘Felina 34’ was high in alpha-terpinolene
and the fiber cultivar ‘Ferimon 12’ had a large
caryophyllene oxide concentration. Drug types were generally lower in
caryophyllene oxide content. The best fragrance rating ("quite good")
was ‘Felina 34’, and the one with the least favorable rating ("quite
bad") was ‘Fedora 19’ (Tab. 2).
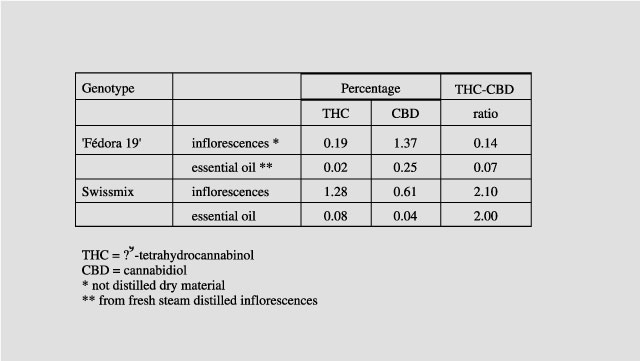
THC
concentration in the essential oil was very low, even in drug varieties,
reaching 0.08% in Swissmix (Tab. 3). THC concentration was lower and the ratio
of THC to cannabidiol was not higher in the essential oil compared to the
inflorescences.
Table 2. Chemical composition of essential oils of some Cannabis sativa L. strains.
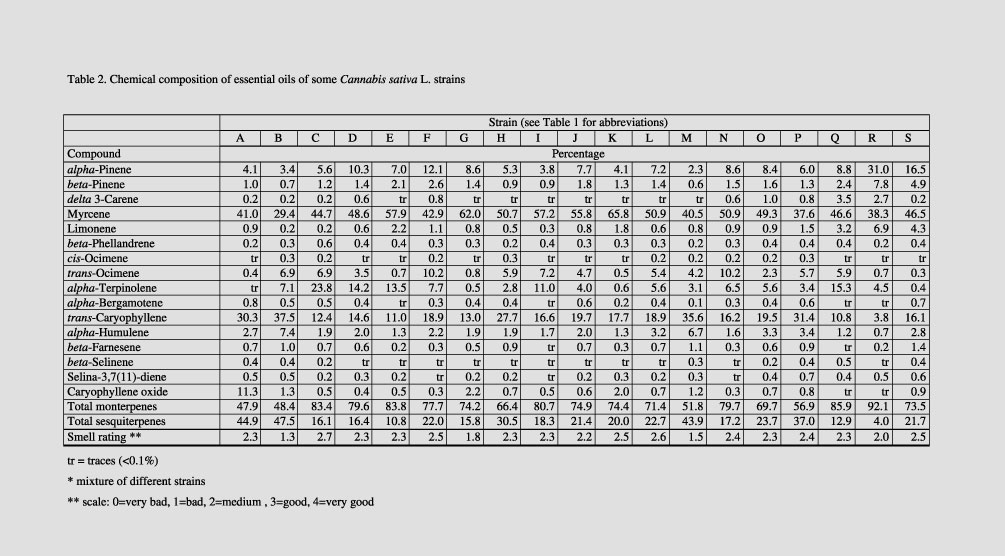
Table 3. Cannabinoid concentration in female or hermaphrodite inflorescences and in the essential oil of two Cannabis strains.
Discussion
The characterized compounds are the major constituents of
hemp essential oil as described by Hendriks et al.
(1975), Turner et al. (1980) and Ross and ElSohly (1996). Because of its
low volatility and water insolubility (Malingré et al. 1975), THC
concentrations in the essential oils were low. Therefore, the use of this steam
distilled oil for drug purposes is not expected.
Smell
is, of course, a very subjective phenomenon. For that reason, smell test ratings
varied considerably. Oils with high sesquiterpene concentrations received a low
rating, meaning that they smelled badly. In contrast, oils with high monoterpene
percentages (but a low alpha-humulene or caryophyllene oxide
concentration) got a high rating. Surprisingly, a mixed oil from different
strains received the best rating. This could be an important consideration for
future commercial use.
These
preliminary results must be interpreted with caution. Harvest stage and the
weather "just-before-harvest" may influence the quality of this
essential oil, which could be developed into a promising product for the
cosmetic, food, medical and plant protection sectors.
References
Acknowledgments
We thank I. Slacanin (Laboratoire Central, Bienne) for the
analyses of monoterpenes and sesquiterpenes and R. Brenneisen (Institute of
Pharmacy, University of Bern) for the analyses of cannabinoids.